The potentially protective role of lactoferrin in atherosclerosis
Vascular calcification
Duration: 4 minutes

The potentially protective role of lactoferrin in atherosclerosis (vascular calcification)
Many cardiovascular diseases, such as stroke, hypertension, heart failure, venous disease, as well as coronary heart disease, which can lead to myocardial infarction, can be traced back to the clinical picture of atherosclerosis.[1]
Atherosclerosis develops in association with endothelial cell dysfunction, smooth muscle cell proliferation and migration, cholesterol accumulation, chronic inflammation, foam cell formation, atheromatous plaque formation, and eventually leads to thrombus formation.[2]
Cardiovascular diseases are a major cause of morbidity and mortality worldwide and represent an immense health but also economic burden.
It is therefore important to exploit and promote preventive options at an early stage.[3]
Lactoferrin can be a significant factor here at 3 points:[4]
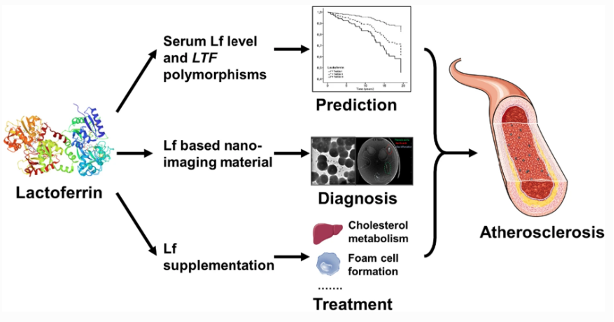
Physiological Lf is associated with the risk of atherosclerosis. Epidemiological studies found that endogenous Lf levels are also associated with the severity of coronary stenosis and the risk of cardiovascular events.[5]
In one clinical study, blood Lf levels were shown to be higher in patients with significant coronary stenosis than in patients without significant coronary stenosis.[4] Another study showed that higher circulating Lf levels were associated with a higher risk of fatal ischaemic heart disease.[5]
In addition, research suggests that circulating Lf levels and specific Lf gene polymorphisms are associated with atherosclerosis risk and that Lf may be a novel marker for atherosclerosis; however, the predictive value of Lf in atherosclerosis and atherosclerosis-related disease has yet to be verified in a large-scale study.[6]
In addition, Lf has also been used in the synthesis of contrast agents for magnetic resonance imaging (MRI) to detect AS.[4]
Several clinical studies showed that Lf administration is involved in several mechanisms of risk factors for the development and progression of AS:
Improved cholesterol metabolism
Bovine Lf (bLf) can increase the "good cholesterol", the HDL (high-density lipoprotein) level in plasma and at the same time reduce the level of "bad cholesterol and fats", i.e. triacylglycerides, cholesterol, non-esterified fatty acids and hepatic cholesterol. The underlying mechanism is not yet known.[7]
Cholesterol homeostasis may also be improved by bLf. This is regulated in the liver and small intestine, where the role of hepatocytes is to convert cholesterol into bile acids. These are subsequently absorbed by epithelial cells in the small intestine. Animal studies have shown that bLf can promote this process by excreting significantly more bile acids and, in parallel, lowering cholesterol levels in the liver and alleviating symptoms of AS.[8,9]
In summary, bLf may improve cholesterol metabolism by reducing cholesterol synthesis and absorption and increasing cholesterol excretion.
Inhibition of foam cell formation
Foam cells arise from macrophages (so-called "scavenger cells") that uncontrollably ingest cell-modified LDL (low-density lipoprotein) and accumulate cholesterol esters, among other things. BLf may be able to interact with cell-modified LDL, specifically acetyl-LDL, and neutralise its negative charge, thus inhibiting binding to macrophages and reducing foam cell formation.[10]
Binding to advanced glycation end products (AGEs)
AGEs can promote the production of reactive oxygen species (ROS) (promoting oxidative reactions), activate the NF-κB signalling pathway and stimulate the expression of VCAM-1.[4]
One study found that Lf can bind to AGEs and interact with AGE ligands, affecting subsequent intracellular signal transduction. This finding suggests that Lf may be indirectly involved in the pathogenesis of atherosclerotic disease by responding to AGEs.[4]
Lowering homocysteine and leptin levels
The amino acid homocysteine is an important marker in the blood for the progression of AS, as an elevated level is associated with AS lesions and accelerated plaque formation, while a lowering of the marker can have a preventive effect against cardiovascular disease.[4]
Leptin, in turn, is produced by adipocytes ("fat stores") and stimulates vascular inflammation and oxidative stress.[4]
Clinical animal studies confirm that oral ingestion of bLF can significantly reduce both markers, which may be an important mechanism for the effect of Lf on AS.[11]
Antiinflammatory function
Lactoferrin is known for its anti-inflammatory effect. Inflammation, in turn, plays a central role in AS. A number of inflammatory factors (monocyte chemoattractant protein-1, IL-8, ICAM-1, VCAM-1, selectin) act as attractants for various immune cells that bind to the endothelium of arterial walls and trigger inflammatory responses. Numerous studies report that bLf could inhibit excessive inflammatory responses through various immunomodulatory actions.[12]
Antioxidant function
Oxidative stress is triggered by an imbalance between ROS generation and antioxidant defence systems. Increased oxidative stress can trigger inflammatory responses, oxidation of LDL cholesterol, endothelial dysfunction and collagen deposition, and therefore contribute significantly to the development and progression of AS.[4] The extensive antioxidant effects of bLF have already been described in detail in an early news article: Mercurius Production (mercurius-production.com)
These research results show promising approaches for further research and innovative therapeutic approaches.